| Active Ingredient | CHOLIC ACID |
|---|
| Drug Name | FDA Application No. | Company | Dosage Form;Route | Strength | RLD Strength | Original Approval or Tentative Approval Date |
Exclusivity Expiration (NCE) |
Exclusivity Expiration (ODE) |
Chemical Type |
Review Classification |
Marketing Status |
TE Code |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CHOLBAM | (NDA) 205750 | RTRX | CAPSULE;ORAL | 50MG, 250MG | 250MG (RS) | March 17, 2015 | March 17, 2020 | March 17, 2022 | 1 New molecular entity (NME) | P Priority review drug O Orphan drug | Prescription | None |
| Parameters | Details |
|---|---|
| Structural Formula |
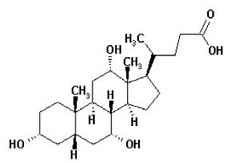 |
| Chemical Name | Cholan-24-oic acid or (3 α, 5, 7 α, 12 α)-3α, 7α, 12α -trihydroxy-5β-cholan-24-oic acid (Cholic acid is a bile acid produced by the liver where it is synthesized from cholesterol.) |
| CAS No | 81-25-4 |
| Molecular Formula | C24H40O5 |
| Molecular Weight | 408.57 |
| Appearance | a white to cream coloured powder comprised of aggregated polycrystalline particles |
| Solubility | It is practically insoluble in water and in 0.1 M HCl at 20°C and is sparingly soluble in 0.1M NaOH at 20°C. It is soluble in glacial acetic acid, alcohols and acetone. A saturated solution in water at 20°C has a pH of 4.4. |
| Water Solubility | 175 mg/L (at 20 °C) |
| Polymorphism | Sufficient evidence was provided to prove that only one crystal form is obtained by the utilised manufacturing process. |
| pKa (Strongest Acidic) | 4.98 (at 20 °C) |
| pKa (Strongest Basic) | - |
| Log P | 2.02 |
| Identification | FTIR and HPLC |
| Degradation | The overall stability data showed that Orphacol is chemically, physically, microbiologically stable and heat Stable. |
| Hygroscopic | non hygroscopic |
| Photostability study | - |
| Melting Point | 198 °C |
| BCS Class | - |
| Manufacture of API | - |
| Parameters | Details |
|---|---|
| Indications and Usage | Bile Acid Synthesis Disordersdue to Single Enzyme Defects: CHOLBAM is indicated for the treatment of bile acid synthesis disorders due to single enzyme defects (SEDs). Peroxisomal Disorders Including Zellweger Spectrum Disorders:CHOLBAM is indicated for adjunctive treatment of peroxisomal disorders (PDs) including Zellwegerspectrum disorders in patients who exhibit manifestations of liver disease, steatorrhea or complications from decreased fatsoluble vitamin absorption. |
| Dosage and Administration | The recommended dosage of CHOLBAM is 10 to 15 mg/kg administered orally once daily, or in two divided doses, in pediatric patients and in adults. |
| Mechanism of action | Cholic acid is a primary bile acid synthesized from cholesterol in the liver. In bile acid synthesis disorders due to SEDs in the biosynthetic pathway, and in PDs including Zellweger spectrum disorders, deficiency of primary bile acids leads to unregulated accumulation of intermediate bileacids and cholestasis. Bile acids facilitate fat digestion and absorption by forming mixed micelles, and facilitate absorption of fat-soluble vitamins in the intestine. Endogenous bile acids including cholic acid enhance bile flow and provide the physiologic feedback inhibition of bile acid synthesis. The mechanism of action of cholic acid has not been fully established; however, it isknown that cholic acid and its conjugates are endogenous ligands of the nuclear receptor, farnesoid X receptor (FXR). FXR regulates enzymes and transporters that are involved in bile acid synthesis and in the enterohepatic circulation to maintain bile acid homeostasis under normal physiologic conditions. |
| Absorption | Orally administered cholic acid is subject to the same metabolic pathway as endogenous cholic acid. Cholic acid is absorbed by passive diffusion along the length of the gastrointestinal tract. Once absorbed, cholic acid entersinto the body’s bile acid pool and undergoes enterohepatic circulation mainly in conjugated forms. |
| Food Effect | - |
| Distribution | In the liver, cholic acid is conjugated with glycine or taurineby bile acid-CoA synthetase and bile acid-CoA: amino acid Nacetyltransferase. Conjugated cholic acid is actively secretedinto bile mainly by the Bile Salt Efflux Pump (BSEP), and thenreleased into the small intestine, along with other components of bile. |
| Metabolism | Conjugated cholic acid is mostlyre-absorbed in the ileum mainly by the apical-sodium-dependent-bile acid transporter, passed back to the liver by transporters including sodium-taurocholate cotransporting polypeptide and organic anion transport protein and enters another cycle of enterohepatic circulation. Any conjugated cholic acid not absorbed in the ileum passes into the colon where deconjugation and 7-dehydroxylation are mediated by bacteria to form cholic acid and deoxycholic acid which may be re-absorbedin the colon or excreted in the feces. |
| Elimination | The loss of cholic acid is compensated by de-novo synthesis of cholic acids from cholesterol to maintain the bile acid pool in healthy subjects. |
| Peak plasma time (Tmax) | - |
| Half life | - |
| Bioavailability | - |
| Age, gender | - |
| DMF | Status | Type | Submit Date | Holder |
|---|---|---|---|---|
| 27181 | A | II | June 7, 2013 | NEW ZEALAND PHARMACEUTICALS LTD |
| Parameters | Details | ||
|---|---|---|---|
| Strength | 50MG | 250MG | |
| Excipients used | silicified microcrystalline cellulose, magnesium stearate and hard gelatin capsules | ||
| Composition of coating material | - | ||
| Composition of caspule shell | The size 2 shells contain gelatin, red iron oxide and titanium dioxide | The size 0 shells contain gelatin and titanium dioxide | |
| Pharmaceutical Development | The aim of pharmaceutical development was to obtain capsules containing 50 mg or 250 mg of cholic acid when filled by adjustment to the volume of size 3 or size 0 capsules respectively. The particle size of the active substance was important for the volume adjustment of the powder in the empty capsule. As a consequence a particle size specification is set for lactose monohydrate and it is comparable to that of cholic acid. The active substance needs to be milled prior to use as a number of large particles were observed in unmilled active substance which could affect blend homogeneity. | ||
| Manufacture of the product | The manufacturing process is very simple consisting of premixing and mixing stages followed by lubrication and filling of capsules. The main parameters of importance are homogeneity of the final lubricated mix and homogeneity of the capsules. | ||
| Tablet / Capsule Image |

|
||
| Appearance | Size number 2 Swedish orange capsule with cap imprinted with “50mg” and body imprinted with “ASK001”. The capsules contain a white to off-white powder. | Size number 0 white capsule with a cap imprinted with “250mg” and body imprinted with “ASK002”. The capsules contain a whiteto off-white powder. | |
| Imprint code / Engraving / Debossment | imprinted with “50mg” and body imprinted with “ASK001” | imprinted with “250mg” and body imprinted with “ASK002” | |
| Score | no score | no score | |
| Color | Orange | White | |
| Shape | Capsule | Caspule | |
| Dimension | Size 2 (18mm) | Size 0 (22mm) | |
| Mfg by | Patheon France SA (US) | ||
| Mfg for | Asklepion Pharmaceuticals LLC (US) | ||
| Marketed by | - | ||
| Distributed by | - | ||
| Application No. | Prod No | Patent No | Patent Expiration | Drug Substance Claim | Drug Product Claim | Patent Use Code | Delist Requested | Link |
|---|---|---|---|---|---|---|---|---|
| There are no unexpired patents for this product in the Orange Book Database. | ||||||||
| USP Apparatus | Speed (RPMs) | Medium | Volume (mL) | Recommended Sampling Times (minutes) | Date Updated |
|---|---|---|---|---|---|
| II (Paddle) with sinker | 100 | Phosphate Buffer, pH 6.8 | 500 mL for 50 mg capsule; 900 mL for 250 mg capsule | 5, 10, 15, 20 and 30 | March 17, 2016 |
| Label | Link |
|---|---|
| FDA label | Download |
| FDA chemistry review | Download |
| FDA Pharmacology Review(s) | Download |
| FDA Clinical Pharmacology Biopharmaceutics Review(s) | Download |
| FDA BE Recommendation | Download |
| European Public Assessment Report | Download |
| Territory | Brand name / Generic company name | Link |
|---|---|---|
| EU | ORPHACOL | Download |
| UK | ORPHACOL | Download |
| US | CHOLBAM | Download |
| - |
| www.accessdata.fda.gov, www.drugbank.ca, www.ema.europa.eu, www.medicines.org.uk, dailymed.nlm.nih.gov |
